Maximise your health potential! Alkalize your body! – Video Blog
When we eat foods, they either can have an acidic effect or alkaline effect on the body.
I have been teaching the principles of the alkaline diet for the past 4 years at Trupp Cooking School. During this time I have spoken to many of my students who have tried the diet for various conditions and I have been given a very positive feedback. The diet has demonstrated results such as reduction in joint inflammation (its anti-inflammatory), weight loss and has increased energy levels.
It may not be a diet for everyone tho, when we choose a new diet, we always should take into consideration your constitution, age, digestive capacity, ethnic background, time of the year and lifestyle etc.
Today I wanted to introduce you to the alkaline diet concept, and if this makes sense to you and you feel like you may benefit from such dietary regimen, than go ahead and experiment. For more detailed information don’t hesitate to book an appointment with me or come to my Energise & Alkalise cooking class (press HERE).
Why do we need to alkalize?
Alarmingly high numbers of our population are affected by cancer, cardiovascular disease and diabetes, and these numbers are all increasing! This growth correlates with the rise in consumption of acid forming foods such as sugars, Trans fats, fast food, refined food such as white breads. At the same time our consumption of fresh alkaline vegetables and essential fatty acids has decreased dramatically.
Acid–base homeostasis is the part of human homeostasis concerning the proper balance between acids and bases, also called body pH. The body is very sensitive to its pH level, so strong mechanisms exist to maintain it. Outside the acceptable range of pH, proteins are denatured and digested, enzymes lose their ability to function, and death may occur.
The body’s acid–base balance is normally tightly regulated by buffering agents, the respiratory system, and the renal system, keeping the arterial blood pH between 7.38 and 7.42. Several buffering agents that reversibly bind hydrogen ions and impede any change in pH exist. Extracellular buffers include bicarbonate and ammonia, whereas proteins and phosphate act as intracellular buffers; the relationship between multiple buffers in the same solution is described by the isohydric principle. The bicarbonate buffering system is especially key, as carbon dioxide (CO2) can be shifted through carbonic acid (H2CO3) to hydrogen ions and bicarbonate (HCO−3).
Diet can slightly alter the body’s acid-base balance. It is advisable by many nutritionists to eat diet that helps your body maintain its acid-base homeostasis by providing foods rich in minerals, such as plan foods of all kinds, especially the green leafy vegetables.
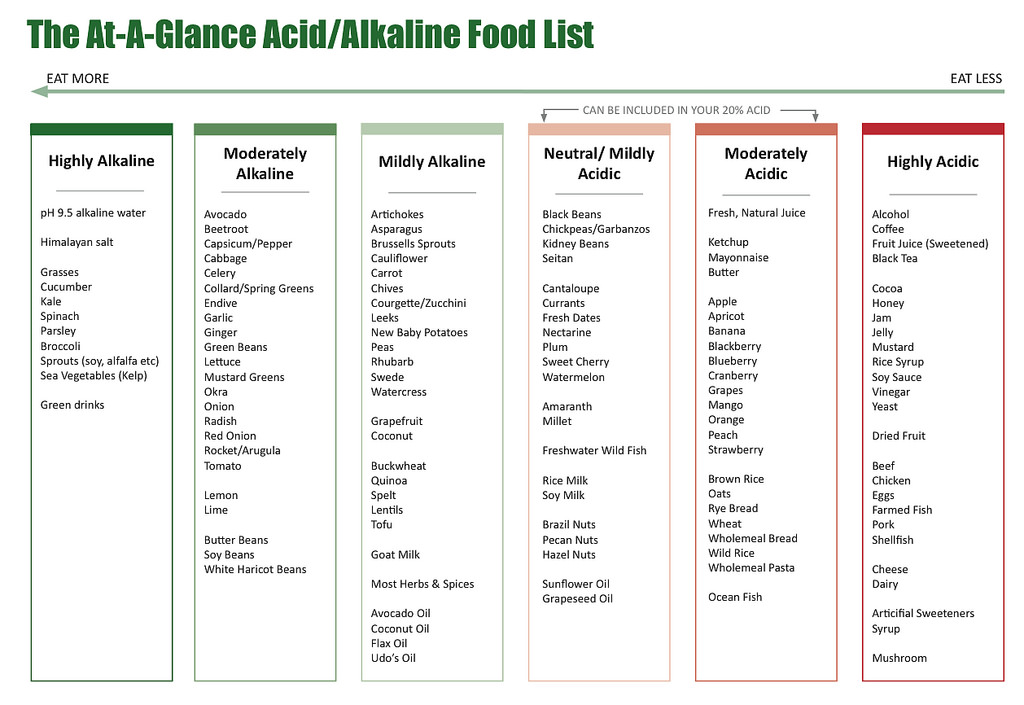
The theory of alkaline diet advises you to consume about 80% alkaline foods and 20% acid foods.
Below is a brief acid/alkaline food list that gives an overview of where food catagories fit into the spectrum.

Acid-alkaline food chart designed by “Energise for life”.